Bovine Brain Damage: New Findings in the Field of Wildlife Neuroscience

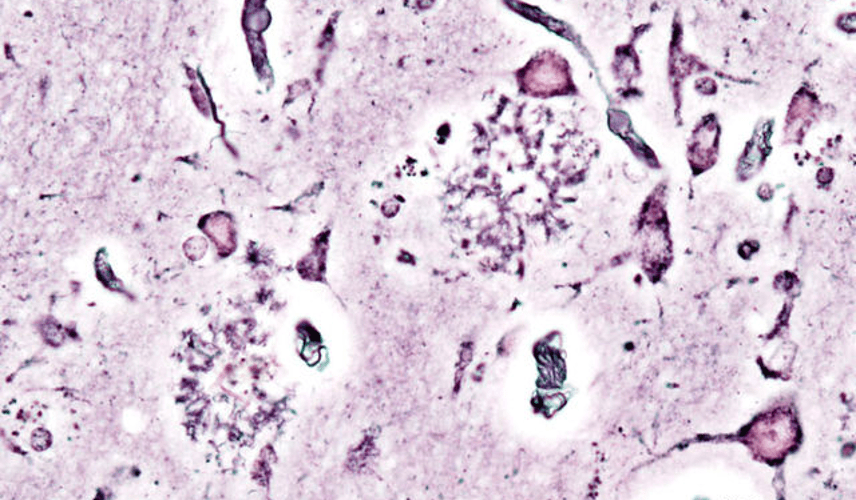
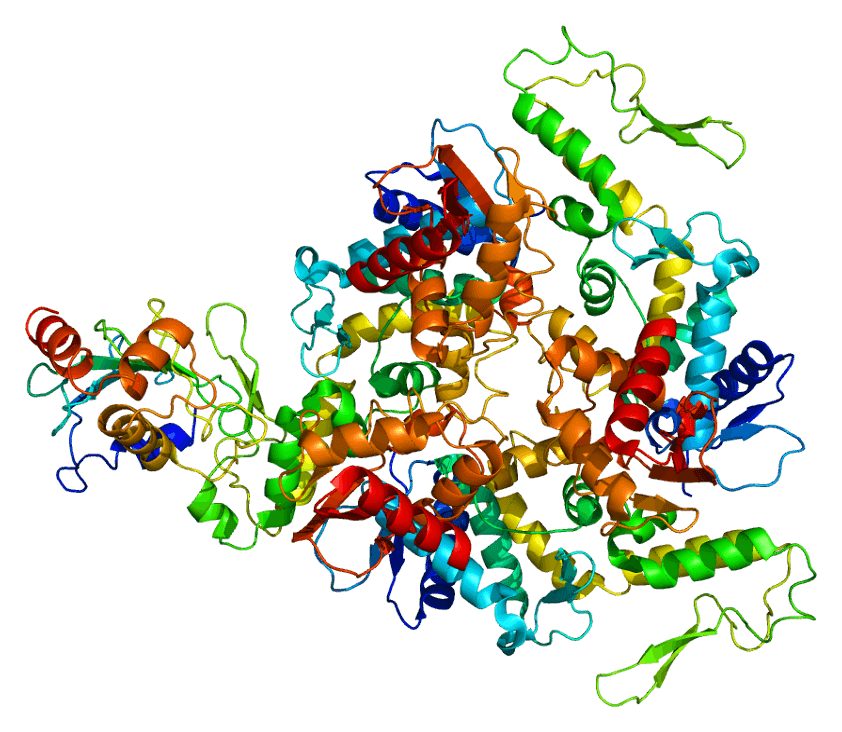
Gretchen Carpenter ’23, Neuroscience, 22F In May 2022, Ackermans et al. from the Icahn School of Medicine at Mount Sinai published an intriguing study, directly contradicting the previous commonly held understanding that humans were the only animals to suffer from chronic traumatic encephalopathy (CTE). The findings? Headbutting muskoxen showed signs … Continue reading