- Home
- Join AIC
- Certification
- Links
- About AIC
- Mission Statement
- Message from the President
- Officers
- Board of Directors
- Code of Ethics
- By-Laws
- History of AIC
- Activities
- Annual Meeting
- Meeting Minutes
- Local Institutes
- NEIC
- Members and Fellows Lecturers
- Presidential Citations
- News
- Announcements
- National Meeting
- News Feeds
- Environment News
- Science News
- Chemicals Industry News
- Top US News
- Awards
- AIC Gold Medal Award
- Chemical Pioneer Award
- Student Awards
- Ethic Awards
- Photos
- THE CHEMIST - Official Journal
- Opportunities
- Career Services
- Message Boards
- Job Postings
- Volunteer Opportunities
- Outreach
- International
- Contact Us
- AIC President
- Board Members
The Determination of Cefadroxil by Using Organic Reagent NQS
Hind AbdulWhaab Abdulateef 1 , Dheyaa H. Ibrahim 1 , Zaid H. Mahmoud *2
1 Department of Chemistry, College of Education for Pure Sciences University of Diyala, Iraq
2 Department of Chemistry, College of Sciences University of Diyala, Iraq
(E-mail: hhindabd@gmail.com , dheyaahus@gmail.com )
* Corresponding author (E-mail: zaidhameed_91@yahoo.com )
Abstract:A simple spectrophotometric method to determine the cefadroxil has been investigated. The method is based on Schiff's base reaction of cefadroxil with 1,2-naphthoquinone-4-sulfonic acid (NQS) in an aqueous solution to give a dark yellow product for maximal absorption of 460 nm. It complied with Beer's Law with a range of 0.1 – 20 µg/ml. The limit of detection (LOD) and the limit of quantitation (LOQ) were 0.0339 and 0.169 ppm, respectively. It was found that the average recovery percent was 99.89% and 1:1 product. As the stability constant was 2.7 × 10 6 L/mol, this method was successfully applied for the determination of cefadroxil in the pharmaceutical formulations. The results obtained using this method are compatible with the method of the British Pharmacopoeia method.
Key Words:cefadroxil, Shiff's base
Introduction
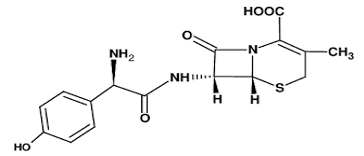
Cefadroxil is an antibiotic [1] consumed and used to treat mild to moderate infections caused by susceptible microorganisms [2]. It is used to treat bacterial infection of the skin and strep throat for the urinary tract [3,4]. Figure 1 shows the structure of the drug.
 |
|---|
|
Several scientific methods of analysis were available in the literature for the determination of cefadroxil in their pharmaceutical preparations, including fluorimetry [6] and polarography [7,8]. Thin layer chro- matography [9], HPLC [10,11], sequential injection analysis [12], chemiluminescence [13,14], capillary electrophoresis [15], and spectrophotometric methods have been described to determine cefadroxil using various reagents based on the formation of complexes with copper (II) [16] Flow injection analysis (FIA) [17]. Additionally, another method is based on the liberation of hydrogen sulfide and followed by the reaction with N,N-diethyl-p-phenylenediamine [18].
Other spectrophotometric methods are reported for the determination of cefadroxil based on its reactivity with iodine [19]. Nitrosation and subsequent metal chelation reaction with 2,6-dichloro-quinone-4-amino-antipyrene in the presence of potassium hexacyanoferrate [20] or by oxidation in an acid medium [21].
These methods are time-consuming and required extraction steps or required indirect procedures. This work describes a simple and sensitive spectrophotometric method for the determination of cefadroxil. This method is based on the reaction of the drug with NQS and the formation of Schiff's base.
Experimental Apparatus
- Spectrophotometer using Ajena model 1100 (Germany) with a quartz cell with 1 cm path length, PW “9421” pH meter for a common glass electrode.
A meter electrical balance was used to weigh the sample. The reagent was supplied by BOH and Fluka. The standard solution of 100 ppm cefadroxil was prepared by dissolving 0.01 g in 2 ml of de-ionized water and then diluted to 100 ml. Also, 5 × 10-3 M of 1,2-naphthoquinone reagent was made by dissolving 0.06 g in 50 ml de-ionized water.
Results and Discussion
In a primary test, the NQS reagent reacted with cefadroxil in the presence of sodium hydroxide NaOH and formed a red color product with the highest absorption peak at 465 nm, where the reagent blank showed low absorbance at this wavelength.Study of Optimal Reaction Conditions
Impact of NQS Reagent
The impact of changing the reagent 1,2-naphthoquinone solution concentration onthe absorbance of cefadroxil was performed. It was noticed that the absorbance increasedand reached a maximum when 1 ml of 5 ×10 -3 M 1,2 - naphthoquinone solution wasused (Figure 2).
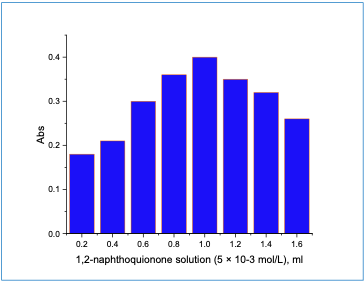 |
|---|
|
Impact of Base
The impact of bases (sodium hydroxideNaOH, potassium hydroxide KOH,ammonium hydroxide NH 4 OH, and sodiumcarbonate Na 2 CO 3 ) was investigated. It wasfound that potassium hydroxide gave maximum absorption at 460 nm (Figure 3).
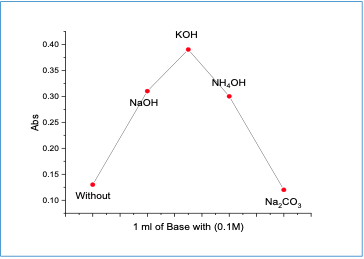 |
|---|
|
Furthermore, the impact of the potassium hydroxide volume and pH were studied. Maximum absorbance was observed when 1.5 ml of 0.1M KOH at pH 11.31 was used.
Table 1. The Impact of Increasing pH and the Volume of KOH 0.1 M on the Absorption of the Mixture (8 ppm Cefadroxil, NQS, KOH) |
 |
Impact of Surfactants
The impact of Tween 80 (TW-80), Triton X- 100 (TX-100), and cetyltrimethyl ammonium bromide (CTAB) of 0.1% concentration was studied. However, the absorbance was decreased when CTAB was used (Figure 4). Therefore, it was excluded from the experiment.
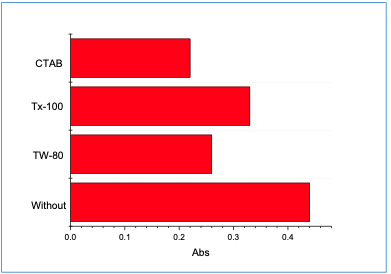 |
|---|
|
Impact of Temperature Versus Time on the Absorbance of the Complex
The effect of reaction time was performed at different temperatures. Figure 5 shows a decrease in the absorbance when time increased, attributed to the dissociation of the complex. It was found that the optimum time and temperature for the complex was 15 min at 40°C, respectively.
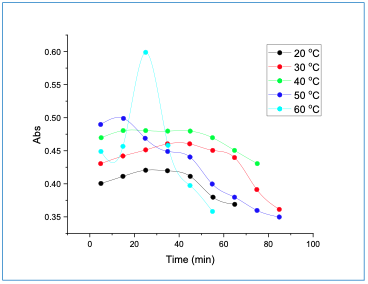 |
|---|
|
 |
|---|
|
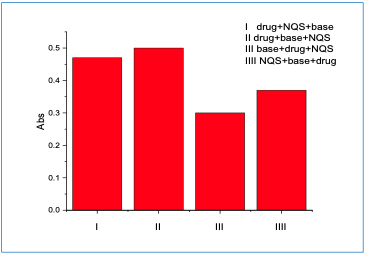
Impact of the Order of Addition
Under the optimum conditions, the order of addition was investigated. Figure 6 shows that in the order of addition, no. II was the best.
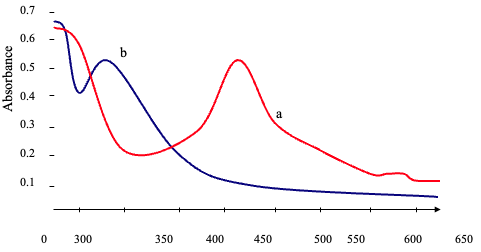
Absorption Spectra
Figure 7 shows the absorption spectra for the best condition that has been confirmed above.
|
Wavelength (nm) |
|---|
Figure 7. (a) The absorption Spectra of Cefadroxil with 1,2-Naphthoquinone Versus Reagent Blank at 460nm (b) Reagent Blank Versus Distilled Water |
The Details of the Statistical Data and Optical Characteristics of the Suggested Method
The absorbance of the complex was measured at 460 nm. Beer's law limits and molar absorptivity values are shown in Table 2. In addition, the relative standard deviation (RSD) and the accuracy of analysis on six replicates for three different concentrations of cefadroxil indicate that the method is valid. Also, the limit of detection (LOD) is accepted as well.
|
 |
Analytical Implementation
The results showed that the experimental F-Test and T-Test were less than the theoretical value (t = 2.50, f = 6.41). However, it was observed that there was no significant variation between the suggested method and the formal method [22].
Quantities and Stability Constant
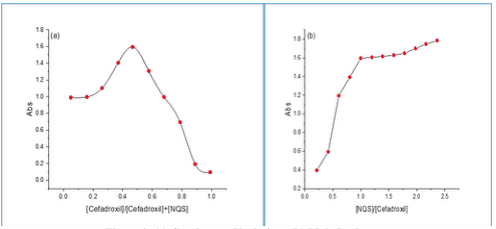
Quantities of a reaction of cefadroxil for NQS were studied through the molar ratio as well as job method [23,24].
|
|
|---|
|
Figure 8 (a) and (b) showed that the results were 1:1 and the average conditional stability constant for the resulting complex was calculated using the equation (1) below:
where K st : the stability constant (L/mol), (∝): the dissociation degree, and (C): the concentration of the resulting complex. The average K st is 2.7 × 10 6 which illustrates that the resulting product is stable.
Mechanism of the Reaction
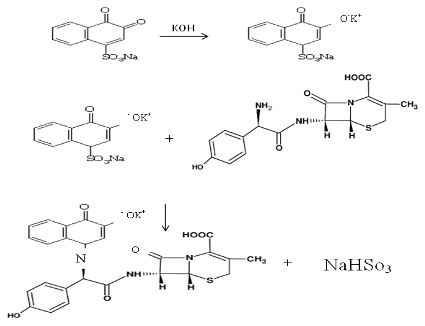
Under the experimental conditions, the mechanism of the reaction is shown in Scheme 1.
|
|
|---|
|
The mechanism suggests that the NQS was converted into a quinoidal which reacts with phenol amine via the replacement of the hydrogen atom of the primary aromatic amine group to produce paraquinoidimide- condensation (Schiff's base) NaHSO 3 .
Conclusion
The suggested spectrophotometric method is simple, sensitive, and low cost. In addition, this method does not involve a solvent extraction step. Also, it gives accurate and precise results. The calibration curve shows high linearity. The coefficient correlation was higher than 0.99. The limit of detection and limit of quantitation values were very acceptable as well. Finally, the suggested mechanism of the product formation shows the NQS was converted to a quinoidal that reacts with phenol amine to produce paraquinoidimide-condensation (Schiff's base) and NaHSO 3 .References
- Buck RE, Price KE. Cefadroxil, a new broad-spectrum cephalosporin. Antimi crob. Agents Chemother. , 1977, 11, 324-330.
- Abdulrahaman AA, Metwally FH, Al-Tam ASI. Spectrophotometric assay of certain cephalosporins based on formation of ethylene blue. Anal. Lett. , 1993, 26, 2619-2635.
- Badawy SS, Abdul-Gawad FM, Ibrahim MM. Spectrophotometric studies on determination of cefadroxil with copper (II) and vanadium (V) in sulphuric acid medium, Anal. Lett. , 1993, 26, 487-497.
- Anjum A, Shetty SKA, Ahmed M, Sridhar BK, Vijaya KML. Development and validation of RP-HPLC method for the quantitative estimation of cefadroxil monohydrate in bulk and pharmaceutical dosage forms. Int. J. Chem. Sci. , 2012, 10(1), 150-158.
- Devaliya R, Jain UK. Noval estimation of cefadroxil in tablet dosage forms by RP-HPLC. Orient. J. Chem. , 2009, 25(4), 1053.
- Yang J, Zhou G, Jie N, Han R, Lin C, Hu J. Simultaneous determination of cephalexin and cefadroxil by using the coupling technique of synchronous fluorimetry and H-point standard additions method. Anal. Chim. Acta , 1996, 325, 195-200.
- Abdel Gaber AA, Ghandour MA. Polarographic studies of some metal (II) complexes with cephalosporins selected from the first generation. Anal. Lett. , 2003, 36, 1245-1260.
- Özkan SA, Erk N, Uslu B, Yilmaz N, Biryol İ. Study on electrooxidation of cefadroxil monohydrate and its determination by different pulse voltammetry. J. Pharm. Biomed. Anal ., 2000, 23, 263-273.
- Shabadi CV, Shelar BA, Shelab AR. Simultaneous determination of cephalexin acid cefadroxil in pharmaceutical preparation by quantitative thin layer chromatography. Indian Drugs , 1998, 35, 766-770.
- Shinde VM, Shabadi CV. Simultaneous determination of cefadroxil and cephalexin from capsule by reverse phase HPLC. Indian Drugs , 1997, 34, 399-402.
- Kano EK, Serra CHR, Koono EEM, Fukuda K, Porta V. An efficient HPLC-UV method for the quantitative determination of cefadroxil in human plasma and its application in pharmacokinetic studies. J. Liq. Chromatogr. Relat. Technol. , 2012, 35(13), 1871-1881.
- Feng S, Jiang J, Fan J, Chen X. Sequential injection analysis with spectrophotometric detection of cefadroxil and amoxicillin in pharmaceuticals. Chem. Anal. (Warsaw, Pol.) , 2000, 135, 191-196.
- Sun. Y. Tang. YH. Yao H, Zheng XH. "Potassium permanganate-glyoxal chemiluminescence determination of cefadroxil antibiotics: Cefalexin, cefadroxil, and cefazolin sodium in pharmaceutical preparations. Anal. Chim. Acta , 2001, 422, 201-202.
- Thongpoon, C, Liawruangrath, B, Liawruangrath S, Wheatley RA, Townshend A. Flow injection chemiluminescence determination of cefadroxil using potassium permanganate and formaldehyde system. J. Pharm. Biomed. Anal ., 2006, 42(2), 277-282.
- Andrasi M, Buglyo P, Zekany L, Gaspar A. A comparative study of capillary zone electrophoresis and pH-potentiometry for determination of dissociation constants. J. Pharm. Biomed. Anal ., 2007, 44(5), 1040-1047.
- Issopoulos PB. Spectrophotometric determination of cephalexin cephradine, ampicillin and amoxicilling using copper (II) acetate as a completing agent. J. Pharm. Biomed. Anal. , 1988, 6, 321-328.
- Ródenas V, García MS, Sánchez-Pedreño C, Albero MI. Spectrophotometric methods for the determination of cephradine or ceftazidine in human urine using batch and flow-injection procedures. J. Pharm. Biomed. Anal. , 1997, 15, 1687-1693.
- Abdalla MA, Fogg AG, Burgess C. Selective spectrophotometric method determination of cephalosporins by alkaline degradation to hydrogen sulphide and formation of methylene blue. Analyst , 1982, 107, 213-217.
- Sastry CSP, Rao SG, Naidu PY, Srinivas KR. New spectrophotometric method for the determination of some drugs with iodine and wool fast blue BL. Talanta , 1998, 45, 1228-1235.
- Salem H. Selective spectrophotometric determination of phenolic B-lactan antibiotic in pure forms and in their pharmaceutical formulation. Anal. Chem. Acta , 2004, 515, 333-341.
- Sasty CS, Rao KR, Prasad DS. Determination of cefadroxil by three simple spectrophotometric methods using oxidative coupling reactions. Microchim. Acta , 1997, 126, 167-172.
- British Pharma Copoeia, Version 4-CD ROM, Her Majesty’s Stationery Office, 2000, pp 8-33.
- Saurina J, Hemandez-Cassos S, Taular R. Anal. Chem. ,1995, 67, 3722.
- Renny JS, Tomasevich LL, Tallmadge EH, Collum DB. Method of continuous variations: Application of job plots to the study of molecular associations in organometallic chemistry. Angew. Chem., Int. Ed. , 2019, 52(46), 11998-12013.
All rights are reserved. © The AIC 2025.
The American Institute of Chemists, Inc. does not necessarily endorse any of the facts or opinions expressed in the articles, book reviews, or advertisements appearing in The Chemist.
Web site designed and developed by RHWD
The American Institute of Chemists, Inc.
315 Chestnut Street, Philadelphia, PA 19106-2702.
Phone (215) 873-8224 | Fax: (215) 629-5224
E-mail: aicoffice@TheAIC.org
315 Chestnut Street, Philadelphia, PA 19106-2702. Phone (215) 873-8224 Copyright - 2017
Designed and maintained by Roy Hagen Web Design